Procedures
Immunohistochemistry technique
Preparation of fresh frozen sections
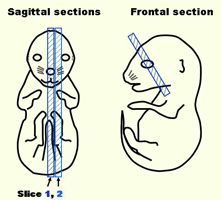
E16.5 whole embryos from pregnant ICR mice were embedded and frozen in OCT compound
(SAKURA Finetechnical, Tokyo, Japan), and stored at -80ºC until use.
Frozen samples were sectioned at 7-10 μm thickness and placed on MSA-coated glass slides
(MATSUNAMI, Japan).
Experiments and animal care have been performed in compliance with
Aichi Medical University and Osaka University institutional animal care.
Standard immunostaining procedures
- Frozen sections were air-dried and treated with fixing reagent*1 at room temperature, and then washed three times with PBS.
- Options: In some cases, sections were treated with a mixture of hyaluronidase and chondroitinase ABC for 30 min at room temperature and/or 0.1M KCl-HCl (pH 1.5) for 0.5-10 min at room temperature before blocking with the serum*2
- Treated sections with 0.3% H2O2/0.3% goat serum*3/PBS for 15 min and then washed three times with PBS.
- Blocked 60 min in 2% goat serum*3/PBS.
- Incubated with primary antibody diluted in 0.1% goat serum*3/PBS.
- Washed four times in PBS.
- Incubated with secondary antibody conjugated with horseradish peroxidase for 60 min at room temperature.
- Washed four times in PBS.
- Added DAB solution to sections and incubated for appropriate time at room temperature.
- Washed with PBS four times.
- Counterstained with hematoxylin by the procedure described elsewhere.
- Dehydrated with graded alcohols and xylene and then applied coverslips with mounting medium.
*1, 2 Conditions for each staining are indicated in the "Antibody information" page.
*3 Calf or fetal bovine serum was used for a goat primary antibody. In some cases, mouse serum was used.
|